Euthanasia drug found in dog food, voluntary recall issued
For most people, pets are seen as other members of the family, which means they care about them just as parents do about their children.
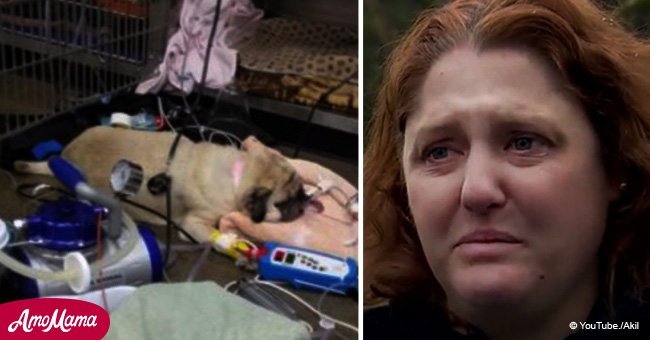
Nikki Mael is one of them. She owned four dogs and all of them were very special for her. One day, she fed them with a can of Evanger’s pet food, what led to her worst nightmare.
Shareably reported that all four dogs fell ill within the next 15 minutes and she took them to the veterinary as soon as possible. Once there, they managed to save three of them but Talula, the fourth one, passed away.
Knowing that the food had something to do with their incident, she sent it to a laboratory to analyze what could have killed Talula.
A couple of days later, the results revealed that the dog food contained pentobarbital, a drug that is regularly used to euthanize dogs, cats, and horses. All of her dogs got poisoned, but Talula wasn’t strong enough to bare it.
More tests were run and they found the same component in Evanger’s and Gravy Train, both produced by Big Heart Pet Foods and owned by J.M. Smucker’s, which is a company that earns around $40 million a year selling pet food.
Even though they didn’t find pentobarbital in the rest of the pet food brands the same company produces, people who buy them got very worried about it. A few of them are Meow Mix, Milk-Bone, Kibbles’nBits, Pup-Peroni, Gravy Train, and Jerky Treats.
J.M. Smucker released a statement apologizing for the incidents and revealed that they identified the cause as a single supplier and a minor ingredient used at one manufacturing facility.
The Food and Drug Administration (FDA) pointed out that pets that eat pentobarbital in small doses can experience drowsiness, dizziness, excitement, loss of balance, nausea, and inability to stand.
If they eat too much of that component, it might cause coma and death. Even though Gravy Train had not enough pentobarbital to be risky, the simple fact that the food has it violates the Federal Food, Drug, and Cosmetic Act.
